Background: Worldwide, the World Health Organization (WHO) estimates that organ transplants currently meet less than 10% of demand. This shortage is even more pronounced for many ethnic minorities, who face significantly lower odds of obtaining suitable matching organs. The preservation of cells, tissues, and organs for transplantation presents unique and scalable opportunities to bridge the gap between supply and demand. The ability to provide organs and tissues on demand has the potential to save or enhance millions of lives globally each year, equivalent to the public health impact of curing cancer. Consequently, our passion for addressing the tissue and organ shortage drives our pursuit of successful biopreservation at the organ, tissue, and cell levels for long-term storage.
Research Line 1: To explore the mechanisms of cryoprotectant (CPA) transport in complicated multivariable biological systems.
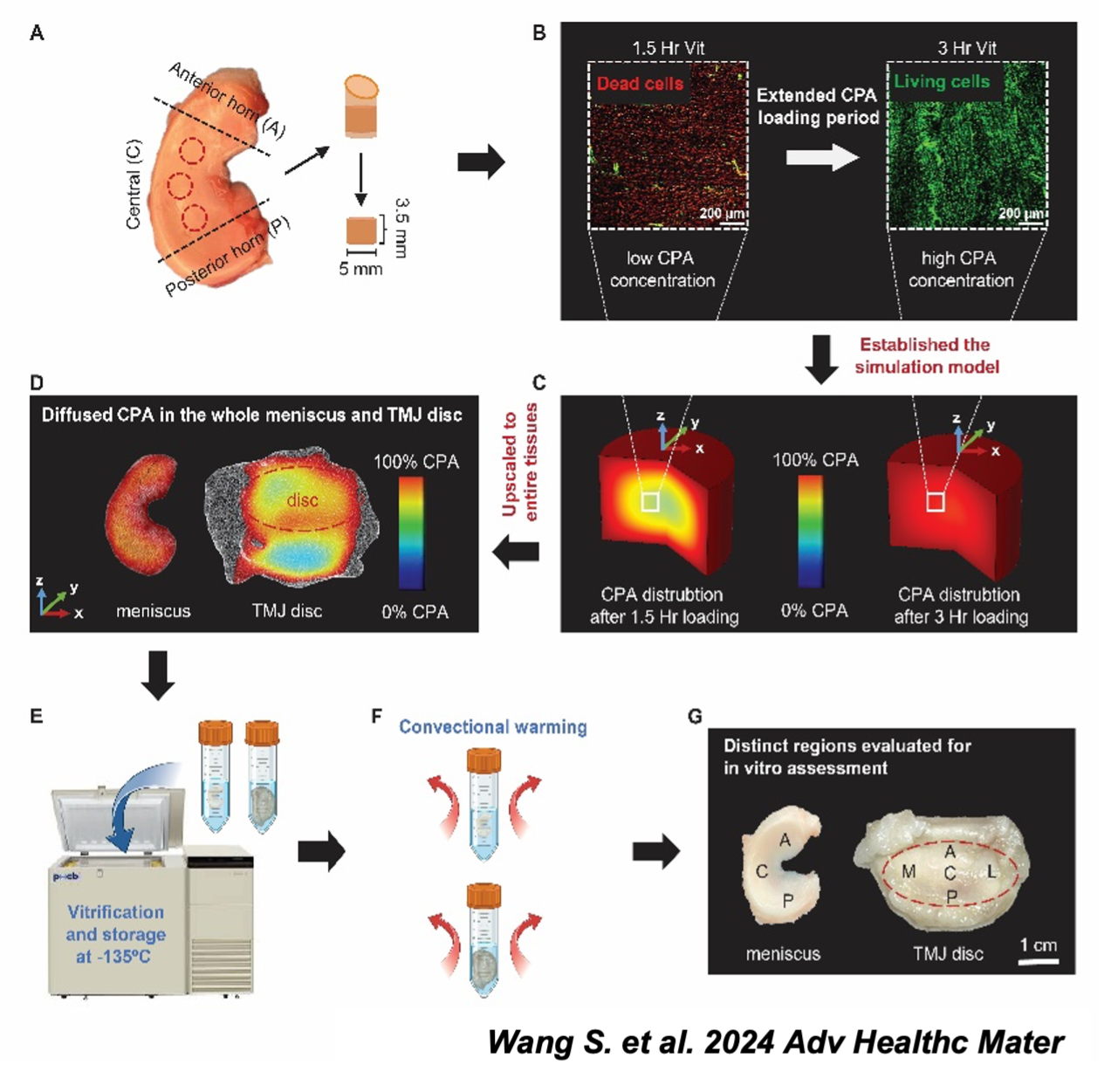
Successful long-term preservation usually requires cryoprotectants (CPAs) to protect cells and tissues from damages caused by ice crystal formation during cooling and warming. To be successful, the CPA needs to penetrate the tissue completely and uniformly. This starts with understanding the permeation kinetics of certain CPAs into the target tissue.
Project 1: Ice-free Cryopreservation with Nanowarming for Banking of Viable Meniscal Transplants [funded by NIH/NIGMS COBRE: SC TRIMH]
Project 2: Ice-free Cryopreservation of Temporomandibular Joint (TMJ) Disc



Research Line 2: To investigate the responses of the biological systems to thermal loading during cooling, warming and/or dehydration.
For cryopreservation via vitrification to succeed, two critical techniques need to be developed: 1) Tissues need to be successfully vitrified (diffusion/perfusion, cooling and storage technologies, and advanced CPAs are needed), and 2) There needs to be a technique that can properly rewarm the tissue for future use. It turns out that the rewarming process is actually the most difficult. Warming rates generally need to be much higher than cooling rates to prevent crystallization, however high warming rates can lead to thermal gradients in the tissue, causing the tissue to fracture. Either crystallization or fracture during rewarming will render the tissue non-viable (useless) for transplant. In this research line, our objective is to pioneer advanced rewarming technologies that facilitate uniform and rapid heating. In addition to the nanowarming technique we developed for Project 1, our collaboration with TDA Research Inc. will lead to the development of an electromagnetic rewarming device utilizing radio frequency (RF) alternating electric fields. This device effectively heats the dipoles in the CPA, enabling rapid and uniform warming of vitrified osteochondral grafts, all without the necessity of nanoparticles.
Project 3: Optimal rewarming solutions for Cryopreserved Tissue Systems [Subaward from TDA Research Inc. funded by DHA]



Research Line 3: To investigate the mechanisms of CPA-induced cytotoxicity and osmotic damage in different biological environment.
Dimethyl sulfoxide (DMSO) is the most common cryoprotectant for cryopreservation of cells and tissues. However, long-term exposure to DMSO has been shown to be cytotoxic when cells are stored above cryogenic temperatures. For most cellular bone graft products, the DMSO cannot be sufficiently removed from allografts during thawing compared to its use in a cell suspension. Therefore, both the allogeneic cell types in the bone allograft and the local cell types at the bone grafting site could be affected as cytotoxicity varies by cell type and by DMSO content. Thus, a DMSO-free cryoprotectant solution is urgently needed. The Project 3 aims to characterize a series of non-toxic cryoprotectants that have been studied to cryopreserve cells in suspension in the cell-seeded bone scaffolds.
Project 4: Development of DMSO-free Preservation Strategies for Long-term Storage of Viable Allografts. [Funded by MTF Biologics]
