THE COTTLE GROUP
Principal Investigator: Dr. Renee Cottle
RESEARCH
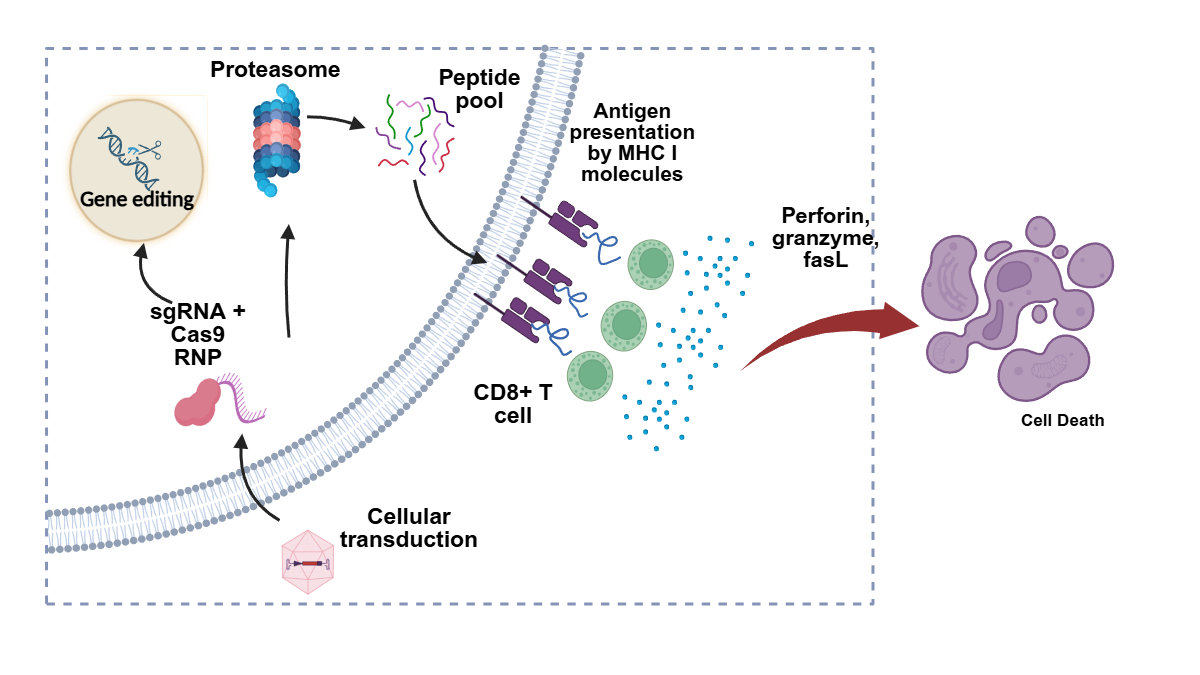
Cas9 immunity and the delivery of CRISPR components into clinically relevant cells are grand challenges in the gene-editing field. Pre-existing Cas9 immunity is prevalent in the human population, with up to 78% of individuals having anti-Cas9 IgG antibodies and Cas9-specific T cells. Anti-Cas9 cytotoxic T cells are particularly problematic: they can eliminate any cell displaying Cas9 peptides on its MHC I molecules. Studies in the literature suggest that AAVs containing SaCas9, when systematically introduced into mice with pre-existing immunity, lead to cytotoxic T cell-mediated elimination of gene-modified target hepatocytes in vivo. It is clear that AAV-mediated delivery of CRISPRs is inhibited by pre-existing Cas9 immunity. Anti-Cas9 cytotoxic T cells can cause serious adverse events, including systemic inflammatory responses. Nonviral approaches allow the delivery of transient, potent forms of Cas9, such as mRNA and ribonucleoproteins, which persist for shorter periods than nucleic acids and are associated with higher gene-editing specificity. Furthermore, ex vivo delivery provides an opportunity to incubate cells until MHC I surface proteins turnover, such that Cas9-derived peptides are no longer expressed on MHC I and cannot trigger T cell responses. Owing to the bacterial origin of Cas9, CRISPR-based therapeutics may elicit host immune responses that compromise patient safety and therapeutic efficacy. The research in the Cottle Lab focuses on strategies to circumvent Cas9 immunogenicity in the context of liver-directed gene therapy. Our research aim is to improve the durability and translational potential of CRISPR-based treatments for inherited metabolic liver diseases.