THE COTTLE GROUP
Principal Investigator: Dr. Renee Cottle
RESEARCH
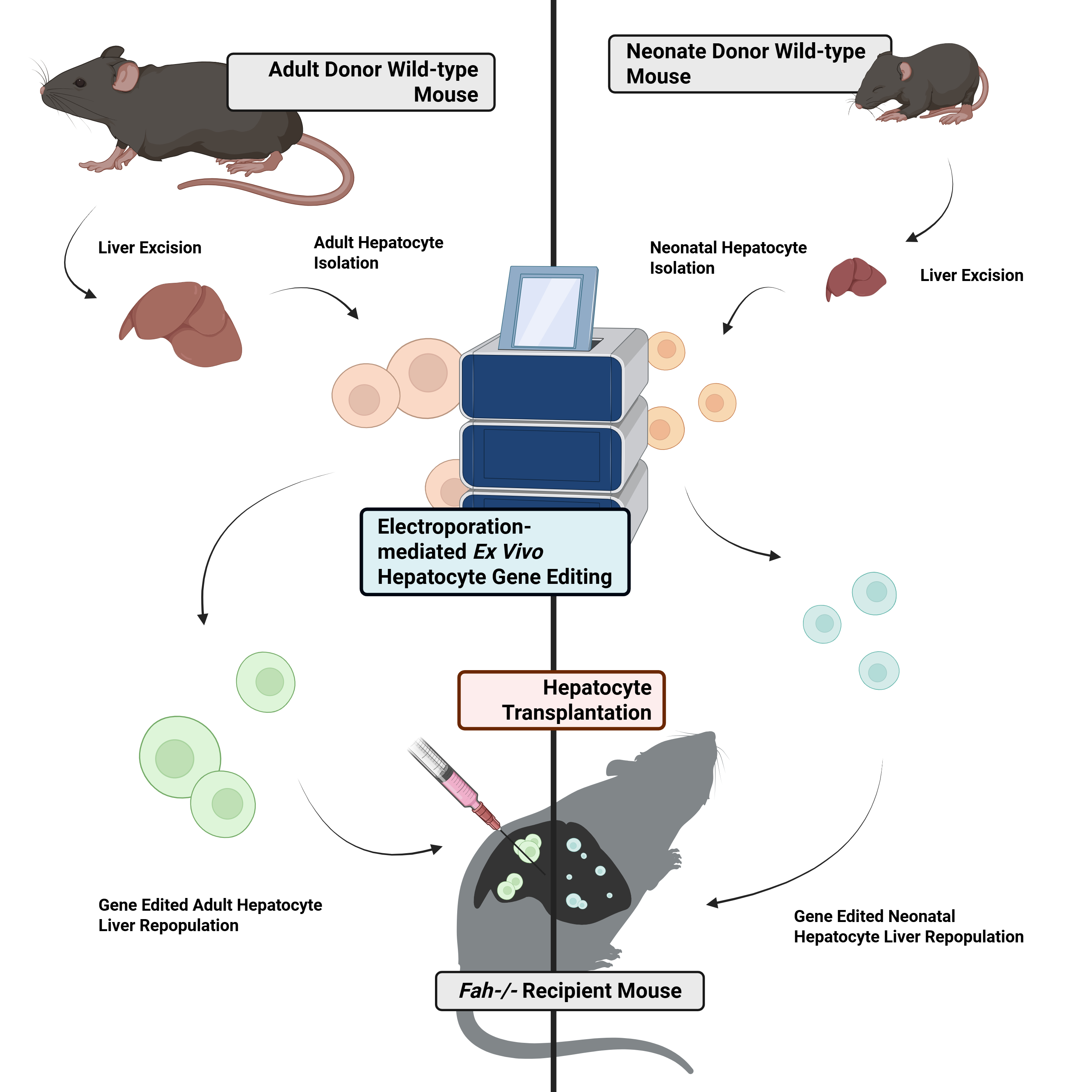
Hepatocyte transplantation (HTx) offers a safer, less invasive alternative to orthotopic liver transplantation for inherited metabolic liver diseases, especially in high-risk pediatric patients. Combining HTx with ex vivo gene editing is a promising autologous therapeutic strategy that uses the patient’s cells. We investigated the feasibility of this approach by applying CRISPR-Cas9-mediated gene disruption in neonatal mouse hepatocytes and comparing their engraftment potential with that of mature adult cells in the Fah-/- mouse model of Hereditary Tyrosinemia Type I. Electroporation-mediated gene editing did not significantly impair the ability of neonatal hepatocytes to engraft in vivo. Quantitative histological analysis revealed comparable liver repopulation levels between recipients of gene-edited neonatal cells and adult cells after hepatoxicity-mediated selection. These findings provide a benchmark for electroporation-mediated gene editing in neonatal hepatocytes and support the development of genetically corrected neonatal hepatocyte products as a crucial long-term or bridge-to-transplant therapeutic strategy for pediatric patients with genetic diseases affecting the liver.