THE COTTLE GROUP
Principal Investigator: Dr. Renee Cottle
RESEARCH
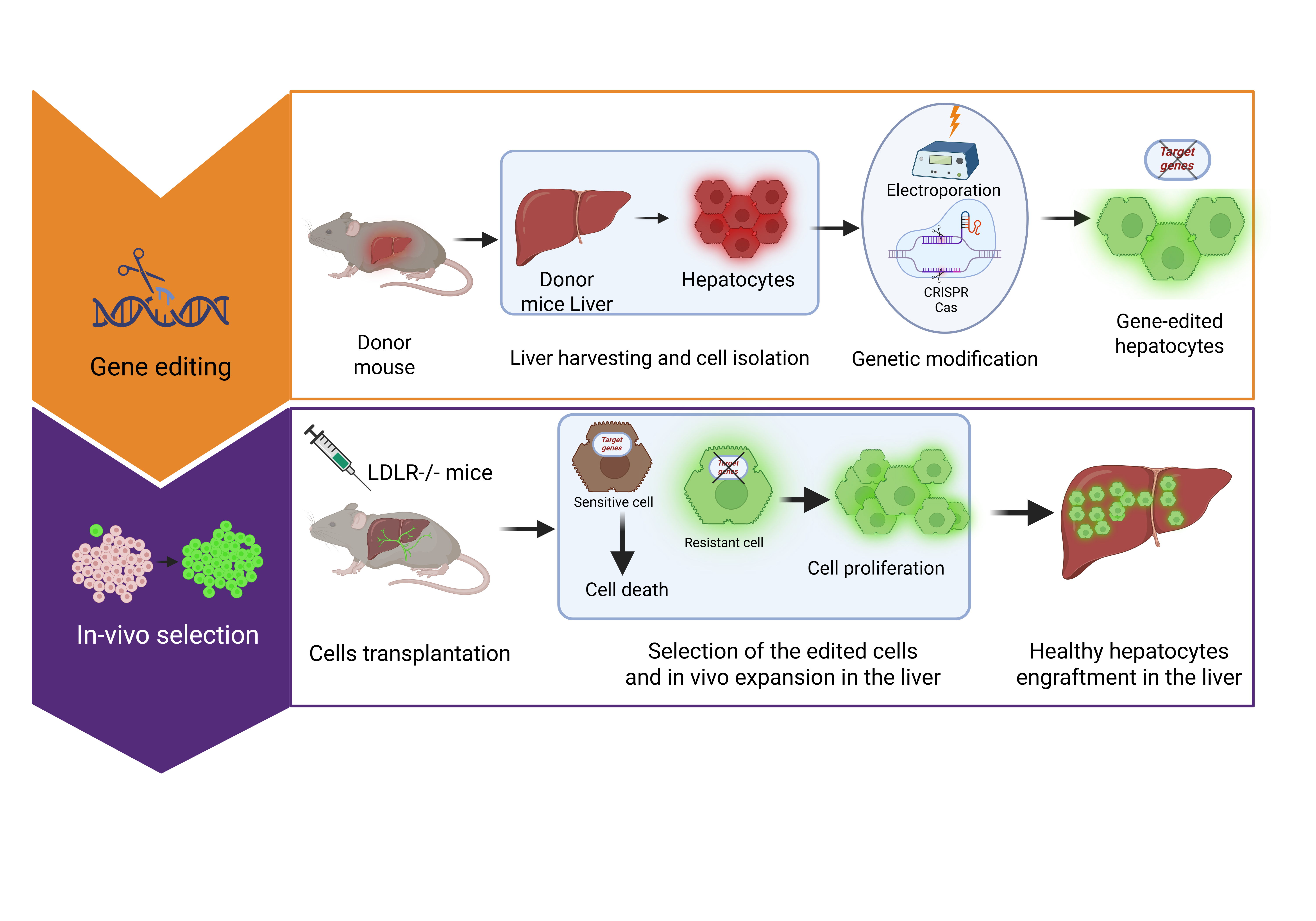 Homozygous familial hypercholesterolemia (HoFH) is an inherited lipid disorder characterized by profound LDL-C elevation, clearance defects, accelerated childhood atherosclerosis, and survival rarely beyond three decades if untreated. LDLR defects account for 90% of HoFH cases by impairing LDL clearance. Current standard therapies, such as statins and PCSK9 inhibitors, upregulate LDLR activity but require residual receptor function. Thus, there is an urgent need for gene therapy, which offers the potential for a one-time, curative approach. Our research employs CRISPR-based gene-editing tools to modify hepatocytes ex vivo via electroporation, followed by transplantation to engraft in the liver and correct the disease. To address challenges in achieving a sufficient engraftment threshold to correct the disease phenotype, methodologies have been developed to confer a selective advantage on gene-edited hepatocytes, thereby facilitating their survival, engraftment, and repopulation within the diseased liver. The Cottle Lab pioneered electroporation of hepatocytes for the treatment of genetic diseases affecting the liver and the development of mouse models. In addition, the Cottle Lab is developing innovative strategies to optimize electroporation in hepatocytes and enhance the engraftment and expansion of gene-edited hepatocytes by precisely inducing hepatotoxicity in native hepatocytes, thereby creating a selective proliferative advantage for transplanted gene-edited cells in vivo. Our research rigorously evaluates the impact of gene editing and selective expansion on liver function, safety, and long-term therapeutic efficacy, while also assessing the overall clinical translatability of these approaches to advance safe, disease-specific cell-based gene therapies for inherited metabolic disease.
Homozygous familial hypercholesterolemia (HoFH) is an inherited lipid disorder characterized by profound LDL-C elevation, clearance defects, accelerated childhood atherosclerosis, and survival rarely beyond three decades if untreated. LDLR defects account for 90% of HoFH cases by impairing LDL clearance. Current standard therapies, such as statins and PCSK9 inhibitors, upregulate LDLR activity but require residual receptor function. Thus, there is an urgent need for gene therapy, which offers the potential for a one-time, curative approach. Our research employs CRISPR-based gene-editing tools to modify hepatocytes ex vivo via electroporation, followed by transplantation to engraft in the liver and correct the disease. To address challenges in achieving a sufficient engraftment threshold to correct the disease phenotype, methodologies have been developed to confer a selective advantage on gene-edited hepatocytes, thereby facilitating their survival, engraftment, and repopulation within the diseased liver. The Cottle Lab pioneered electroporation of hepatocytes for the treatment of genetic diseases affecting the liver and the development of mouse models. In addition, the Cottle Lab is developing innovative strategies to optimize electroporation in hepatocytes and enhance the engraftment and expansion of gene-edited hepatocytes by precisely inducing hepatotoxicity in native hepatocytes, thereby creating a selective proliferative advantage for transplanted gene-edited cells in vivo. Our research rigorously evaluates the impact of gene editing and selective expansion on liver function, safety, and long-term therapeutic efficacy, while also assessing the overall clinical translatability of these approaches to advance safe, disease-specific cell-based gene therapies for inherited metabolic disease.